State-of-the-art pharmaceutical manufacturing plant in Sarigam GIDC, Vapi-Daman Zone, Gujarat with EU-GMP, WHO-GMP, and PICS certifications
State-of-the-art pharmaceutical manufacturing facility in Sarigam GIDC, Vapi-Daman Zone
Globally recognized pharmaceutical manufacturer headquartered near Vapi, Gujarat, operating from a state-of-the-art facility in Sarigam GIDC. Committed to quality and innovation with WHO-GMP, ISO 9001:2018, and forthcoming EU-GMP and PICS approvals.
Strategically located in India's pharmaceutical hub with cutting-edge technology

Taj Pharma Manufacturing Plant - WHO-GMP Certified Facility in Sarigam GIDC, Vapi, Gujarat
Facility: Sarigam GIDC
Zone: Vapi-Daman Zone, Gujarat
Hub: India's generics pharmaceutical center
Advantage: Cost-effective raw materials & skilled expertise
WHO-GMP: Certified manufacturing facility
ISO 9001:2018: Quality management system
EU-GMP: Forthcoming approval
PICS: Pending certification
Tablets: Multiple dosage forms
Capsules: Softgel & hard capsules
Injections: Sterile formulations
Topicals: Creams, ointments & jellies
Extensive product portfolio covering diverse therapeutic categories
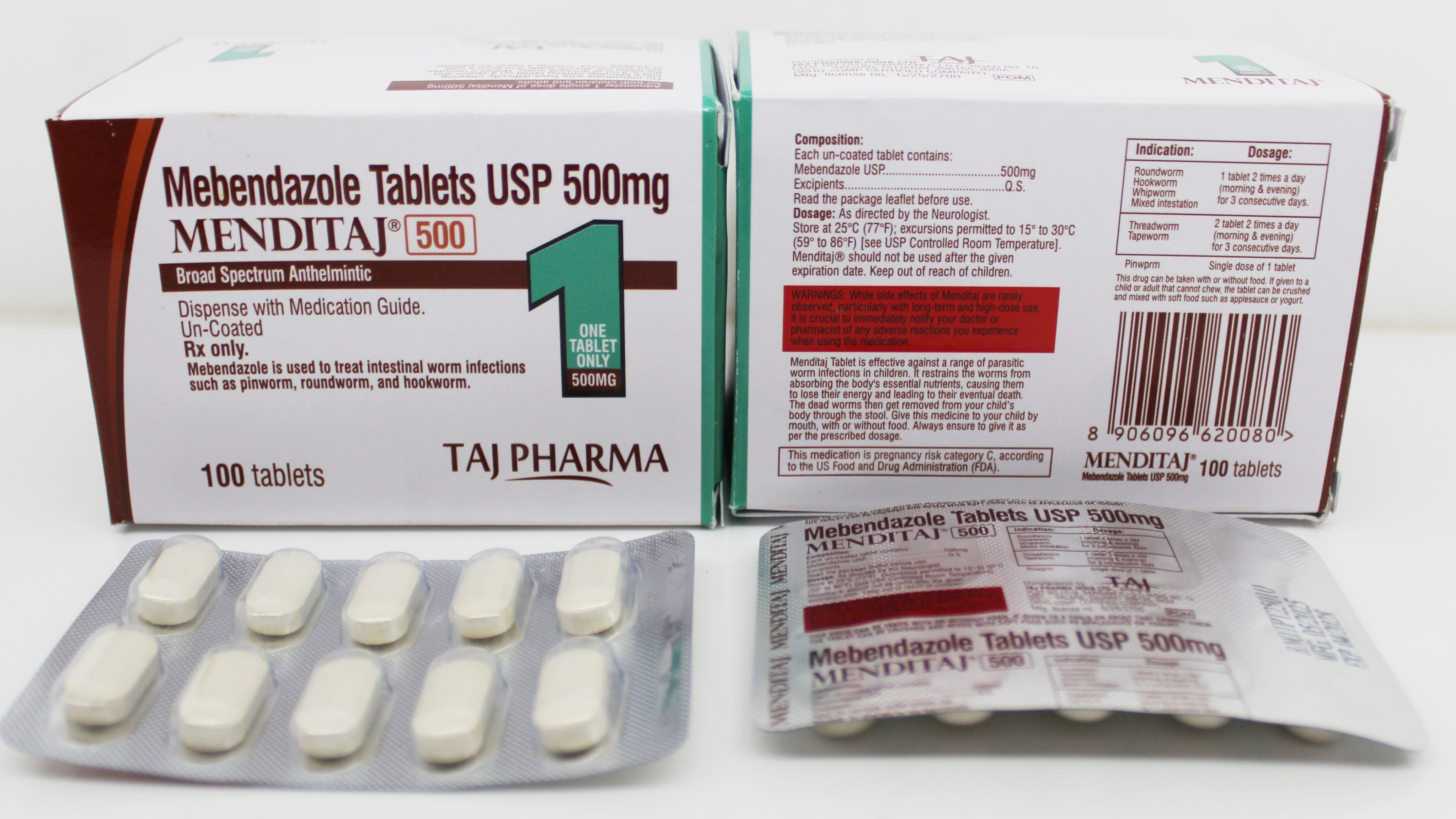
Amlodipine and Valsartan Tablets
Adrenaline Injection
Montelukast Tablets
Progesterone Softgel Capsules
Products distributed worldwide, making quality healthcare accessible to all with competitive pricing and high-quality solutions
Streamlined manufacturing workflow for Mebendazole Tablets 500mg
All incoming raw materials undergo rigorous quality testing including identity, purity, and potency analysis before approval for use.
Wet granulation process using high-shear granulators ensures uniform particle size distribution and optimal flow properties.
High-speed rotary tablet compressors with 30+ stations ensure consistent tablet weight, hardness, and disintegration properties.
Automated vision inspection systems detect defects, ensuring only perfect tablets proceed to packaging.
Automated blister packaging and cartoning in controlled environment to maintain product integrity.
Comprehensive quality testing of finished products including assay, dissolution, and stability testing.
State-of-the-art analytical testing and quality assurance
Controlled environment for pharmaceutical manufacturing
Raw material storage and preparation areas
Granulation and compression areas
Product filling and packaging areas
Aseptic processing and critical areas
Comprehensive quality management system
Global regulatory standards and approvals
Current Good Manufacturing Practices (cGMP) compliance
EudraLex Volume 4 GMP guidelines compliance
WHO-GMP certification and prequalification
Orange Guide GMP standards compliance
Australian GMP Code compliance
PMDA GMP standards compliance
Advanced manufacturing technologies and continuous innovation
Green manufacturing and environmental responsibility
Comprehensive waste segregation and recycling programs with 85% waste reduction
Advanced water treatment and recycling systems with 60% water reuse
Solar power integration and energy-efficient equipment with 30% energy reduction
Advanced emission control systems meeting international environmental standards
Schedule a tour of our WHO-GMP certified manufacturing facility
Our team is ready to provide you with the best pharmaceutical products and support