Certificate of Pharmaceutical Product
Official Certificate of Pharmaceutical Product (COPP) for Mebendazole export and international trade. WHO-GMP certified with complete regulatory compliance for global market registration and distribution.
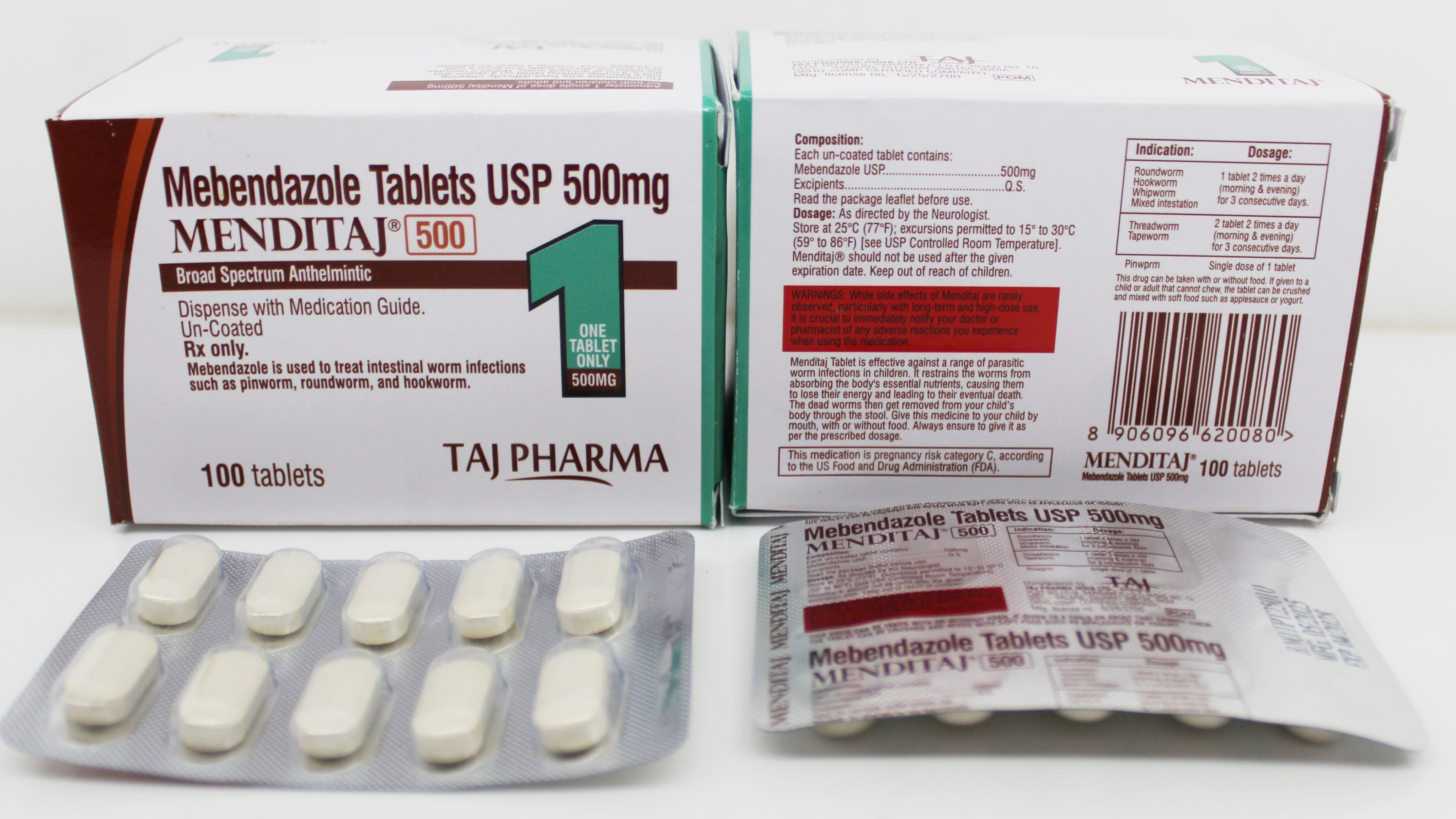
Official Certificate of Pharmaceutical Product (COPP) for Mebendazole export and international trade. WHO-GMP certified with complete regulatory compliance for global market registration and distribution.

The Certificate of Pharmaceutical Product (COPP) is an official document issued by the national regulatory authority that certifies the quality, safety, and efficacy of pharmaceutical products for export. Our Mebendazole COPP certificate is recognized internationally and facilitates smooth customs clearance and market registration.
| Product Name: | Mebendazole Tablets 500mg |
| Brand Name: | Menditaj 500 |
| Dosage Form: | Uncoated Tablets |
| Strength: | 500mg |
| ATC Code: | P02CA01 |
| Therapeutic Class: | Anthelmintic |
| Manufacturer: | Taj Pharma |
| Address: | Vapi, Gujarat, India |
| GMP Certificate: | WHO-GMP Certified |
| ISO Certification: | ISO 9001:2015 |
| PIC/S Member: | Yes |
| Export License: | Valid and Active |
| Issue Date: | Current Date |
| Expiry Date: | 2 Years from Issue Date |
| Renewal Process: | Annual renewal required |
| Update Frequency: | As needed for changes |
| Notification: | 30 days before expiry |
| Global Validity: | Worldwide |
| Regional Recognition: | All major regions |
| Customs Acceptance: | International customs |
| Regulatory Bodies: | WHO, FDA, EMA, etc. |
| Trade Agreements: | WTO compliant |
Prepare all required manufacturing and quality documentation including GMP certificates, product specifications, and stability data.
Submit application to national regulatory authority with complete dossier and supporting documents.
Regulatory authority conducts GMP facility inspection and quality system audit.
Upon successful inspection and review, COPP certificate is issued with validity period.
Obtain your Certificate of Pharmaceutical Product for seamless international export and market registration. Expert support for the entire application process.
Complete Common Technical Document with Module 1-5 structure for regulatory submissions.
CTD DossierWHO-GMP certified manufacturing facilities and quality compliance information.
WHO-GMP InfoComprehensive stability data and shelf life information for all climate zones.
Stability DataOur team is ready to provide you with the best pharmaceutical products and support