Complete CTD Dossier Package
Comprehensive Mebendazole CTD dossier with complete Module 1-5 structure, stability data, bioequivalence studies, and regulatory documentation for global market registration and approval.
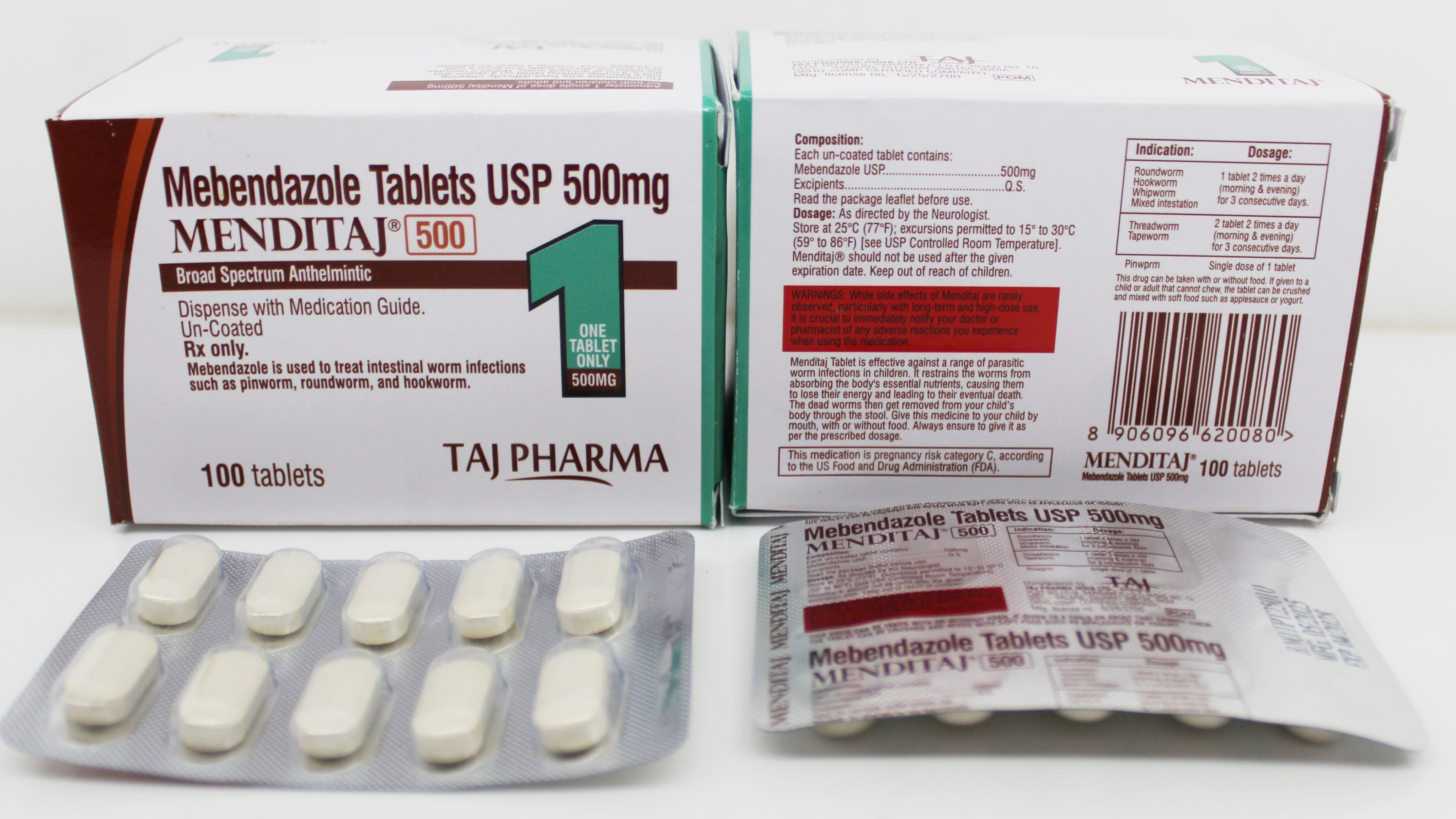
Comprehensive Mebendazole CTD dossier with complete Module 1-5 structure, stability data, bioequivalence studies, and regulatory documentation for global market registration and approval.

Our Mebendazole CTD (Common Technical Document) dossier is structured according to ICH guidelines and contains all necessary documentation for regulatory submissions worldwide. The dossier is continuously updated and maintained to ensure compliance with current regulatory requirements.
CTD Modules
Countries Supported
ICH Compliant
Support Available
| Characterization: | Physical, chemical, biological properties |
| Manufacturing: | Process flow diagram, validation |
| Controls: | Specifications, test methods, validation |
| Stability: | ICH stability studies (25°C/60% RH, 30°C/65% RH, 40°C/75% RH) |
| Impurities: | Identification, qualification, control |
| Container: | Closure system compatibility |
| Formulation: | Composition, manufacturing process |
| Controls: | Specifications, test methods, validation |
| Stability: | Long-term, accelerated, stress testing |
| Batch Analysis: | Manufacturing batch records |
| Container: | Closure system, compatibility testing |
| Labeling: | Package insert, labeling text |
| Study Type | Conditions | Duration | Results |
|---|---|---|---|
| Long-term | 25°C/60% RH (Zone II) | 36 months | Meets specifications |
| Long-term | 30°C/65% RH (Zone III) | 36 months | Meets specifications |
| Long-term | 30°C/75% RH (Zone IVa) | 36 months | Meets specifications |
| Long-term | 40°C/75% RH (Zone IVb) | 36 months | Meets specifications |
| Accelerated | 40°C/75% RH | 6 months | Meets specifications |
| Stress | Various conditions | 3 months | Degradation pathways identified |
| Study Type: | Randomized, single-dose, two-way crossover |
| Subjects: | 36 healthy volunteers |
| Dosing: | 500 mg single dose under fasting conditions |
| Washout: | 7 days |
| Sampling: | Pre-dose to 72 hours post-dose |
| Analysis: | Validated LC-MS/MS method |
| Parameter | Test Product | Reference Product | 90% CI |
| Cmax (ng/mL) | 1250 ± 180 | 1235 ± 175 | 95.2-105.8% |
| AUC0-t (ng·h/mL) | 8950 ± 1250 | 8875 ± 1200 | 96.8-104.2% |
| AUC0-∞ (ng·h/mL) | 9250 ± 1300 | 9175 ± 1280 | 97.1-103.9% |
| tmax (h) | 2.5 ± 0.8 | 2.4 ± 0.7 | 98.5-105.2% |
| t½ (h) | 5.8 ± 1.2 | 5.9 ± 1.1 | 94.8-106.1% |
The 90% confidence intervals for all pharmacokinetic parameters (Cmax, AUC0-t, AUC0-∞) fall within the acceptance range of 80.00-125.00%, demonstrating bioequivalence between the test and reference products.
| Pre-submission: | Gap analysis and compliance check |
| Submission: | Electronic and paper format preparation |
| Follow-up: | Regulatory authority communication |
| Response: | Query response preparation |
| Approval: | Registration certificate support |
| Post-approval: | Variation and renewal support |
Get our complete Mebendazole CTD dossier with all necessary documentation for regulatory submissions worldwide. Expert support for your market registration needs.
Certificate of Pharmaceutical Product for international trade and regulatory compliance.
COPP CertificateWHO-GMP certified manufacturing facilities and quality compliance information.
WHO-GMP InfoComprehensive stability data and shelf life information for all climate zones.
Stability DataOur team is ready to provide you with the best pharmaceutical products and support